VA Hospitals to Administer ANKTIVA® to Bladder Cancer Patients

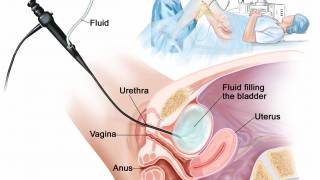
ImmunityBio, Inc. today announced that the Michael E. DeBakey Department of Veterans Affairs (VA) Medical Center recently became the first VA hospital in the Houston, Texas, region and one of the first in the U.S. to provide bladder cancer treatment with ANKTIVA®.
A 2024 study found military exposure to carcinogenic agents was associated with a higher risk for bladder cancer among veterans, making this treatment especially vital for those at the VA.
ANKTIVA, the first of its kind immune-boosting, lymphocyte-stimulating agent, was approved in 2024 by the U.S. Food and Drug Administration (FDA) in combination with Bacillus Calmette-Guérin (BCG) for patients with BCG-unresponsive non-muscle invasive bladder cancer.
BCG vaccination alone has been approved for bladder cancer therapy by the FDA.
By activating NK cells, ANKTIVA overcomes the tumor escape phase of clones resistant to T cells and restores memory T cell activity with resultant prolonged duration of complete response.
Nationally recognized urologic oncologists at the Houston VA, Dr. Jeffrey Jones and Dr. Jennifer Taylor, were instrumental in bringing this novel treatment to veterans.
They are strong advocates for advancing care for those battling bladder cancer.
In addition, Dr. Jones, who is also affiliated with Baylor College of Medicine, led efforts to enroll the facility in ImmunityBio's Expanded Access Program for the recombinant BCG vaccine.
"We are honored to see ANKTIVA reaching our nation's veterans," said Dr. Patrick Soon-Shiong, Founder, Executive Chairman, and Global Chief Scientific and Medical Officer of ImmunityBio, in a press release.
"Drs. Jones' and Taylor's leadership and commitment to innovation are exactly what's needed to expand access to transformative treatments like ANKTIVA across the VA system. This milestone at DeBakey underscores the real-world impact of our mission."
The Company's Cancer Moonshot program was launched in January 2016. Designated an FDA Breakthrough Therapy, ANKTIVA is the first FDA-approved immunotherapy for non-muscle invasive bladder cancer CIS that activates NK cells, T cells, and memory T cells for a long-duration response.
ImmunityBio is applying its science and platforms to treating cancers, including the development of potential cancer vaccines, as well as developing immunotherapies and cell therapies that we believe sharply reduce or eliminate the need for standard high-dose chemotherapy.
As of August 13, 2025, Anktiva plus BCG Vaccine is approved in the UK and the United States and is available at various clinical sites and cancer centers
Our Trust Standards: Medical Advisory Committee