Egypt to Produce Measles-Rubella and Rotavirus Vaccines

Over the past decade, vaccine manufacturers have established production centers closer to where the vaccines are utilized, such as in Africa and Europe.
Today, Batavia Biosciences announced a strategic collaboration with Vaccine Biotechnology City (VBC) and MEVAC to enhance vaccine manufacturing capabilities in Egypt.
As of March 13, 2025, this new collaboration builds on a previously signed license agreement for Measles and Rubella (MR) vaccines. Negotiations for Rotavirus vaccines are underway.
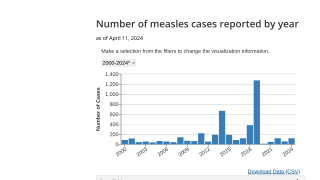
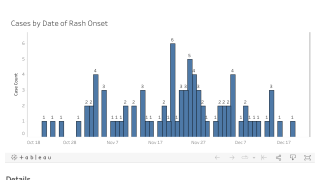
This vaccine production capability is essential during disease outbreaks like measles.
Through this partnership, Batavia will license proprietary processes, analytical methods, and production systems to VBC and MEVAC, empowering them to produce high-quality vaccines locally and meet international standards.
This partnership positions Egypt as a vital player in vaccine manufacturing, contributing to greater vaccine self-reliance for the Middle East and Africa.
In a press release, Ahd Hamidi, Strategic Partnership Director at Batavia Biosciences, stated, “This collaboration reflects our mission to deliver affordable and accessible vaccine solutions to regions where they are most needed. By enabling local vaccine production, we aim to empower Egypt as a critical player in the fight against preventable diseases in Africa and beyond.”
As the collaboration progresses, the Batavia, VBC, and MEVAC partnership is set to make a lasting impact on public health outcomes across Africa and the Middle East.
Our Trust Standards: Medical Advisory Committee