First Approved Chikungunya Vaccine Now Available for European Adolescents

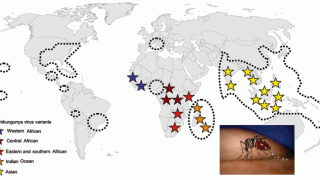
As the global outbreak of the Chikungunya virus continues to expand in India, Brazil, and La Réunion, European adolescents can now receive an effective vaccine that protects them from this mosquito-transmitted disease.
On April 1, 2025, Valneva SE announced that the European Commission (EC) has granted marketing authorization in Europe for Valneva’s single-dose vaccine, IXCHIQ®, for the prevention of disease caused by the chikungunya virus in individuals 12 years of age and older.
With this approval, IXCHIQ® will become available for adolescents in the EU, Norway, Liechtenstein, and Iceland.
In addition to the EC's approval in adolescents and adults in the EU, it has been approved in the United States, Canada, and the United Kingdom to prevent diseases caused by the chikungunya virus in individuals 18 and older. Similar label extension applications for adolescents were also submitted in the U.S., Canada, and the U.K.
Dr. Richard Hatchett, Chief Executive Officer of the Coalition for Epidemic Preparedness Innovations, commented in a media release, “Cases of chikungunya are increasing around the world, making populations of all ages vulnerable to the disease’s long-term debilitating effects, such as prolonged joint pain and inflammation."
"EC’s marketing authorization for use of IXCHIQ® in adolescents in the EU is an important stepping stone that could help accelerate the approval of the vaccine in this age group in other regions, including areas where the disease is endemic.”
In the United States, IXCHIQ is commercially available at most travel clinics and pharmacies and is recommended by the U.S. CDC for people visiting Chikungunya endemic areas in 2025.
Our Trust Standards: Medical Advisory Committee